Rheumatology and Immunology | Prof. Li Zhanguo's Team published a phase II clinical study of the Chinese inovative drug mufemilast in the treatment of Behçet's disease
风湿免疫科栗占国团队在线发表中国原创新药莫米斯特治疗白塞病II期临床研究结果
2026-03-16
Recently, the research team led by Professor Li Zhanguo from the Rheumatology & Immunology Department of Peking University People's Hospital has made another breakthrough in the treatment of Behçet's disease(BD). They published a research paper titled “Efficacy and safety of mufemilast in patients with Behçet's syndrome: a phase II randomized clinical trial” in Annals of the Rheumatic Diseases.

A national multi-center, randomized, double-blind placebo-controlled trial confirmed that mufemilast effectively controls oral ulcers in patients with active BD, significantly reduces pain, and exhibits a safety profile. Behçet's disease(BD), included in the second batch of China’s rare disease catalog, has limited treatment options with traditional drugs. This study offers a new therapeutic choice and marks a key breakthrough in China’s original developed drugs for the condition.
Behçet’s disease is a chronic, recurrent systemic vasculitis, characterized by recurrent oral and genital ulcers, ocular and skin lesions, and potential involvement of joints, blood vessels and other systems. Traditional treatments such as colchicine and thalidomide have a delayed onset of efficacy, a high incidence of adverse reactions and low remission rates, leaving an urgent clinical need for safer, more effective drugs.
During the 12-week placebo-controlled period, the oral ulcer AUC (area under the curve) was significantly lower in the treatment groups than in the placebo group (mufemilast 45 mg group: mean ± SD, 103.9 ± 18.8; least-squares mean difference, −104.8 [95% CI, −156.3, −53.2], P < .001; and mufemilast 60 mg group: 66.1 ± 17.7; least-squares mean difference, −142.5 [95% CI, −192.4, −92.7], P < .001),achieving the primary endpoint (Figure 1).
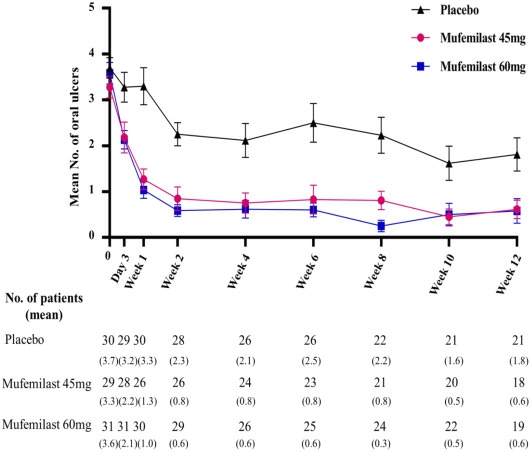
Figure 1. Change in oral ulcer numbers from baseline to week 12
For secondary endpoints, the mufemilast groups showed significant improvements in oral ulcer pain scores (Figure 2) and time to ulcer-free remisson (Figure 3) (both P < 0.001).
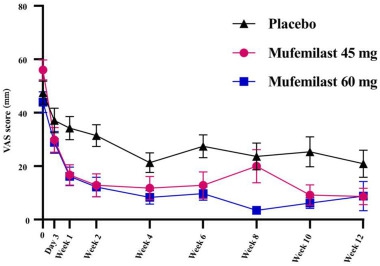
Figure 2. Change in oral ulcer pain from baseline to week 12
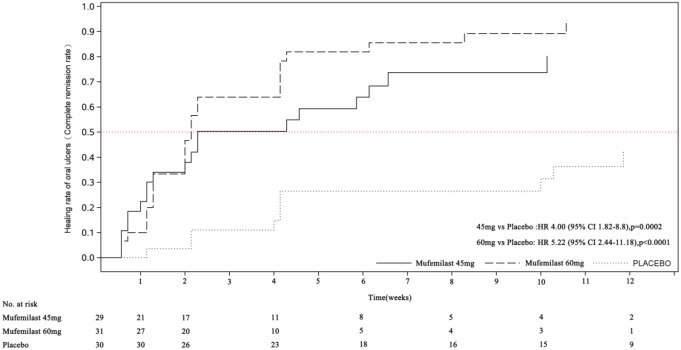
Figure 3. Time to achieve oral ulcer-free remission during the placebo-controlled period
In terms of safety,treatment-related adverse events were predominantly mild and consisted of PDE4 inhibitor-associated gastrointestinal symptoms (e.g., nausea, diarrhea, vomiting), which resolved spontaneously without special treatment. No drug-related serious adverse events were reported throughout the study.
This study confirms that mufemilast (45 mg/d and 60 mg/d), a novel Chinese-developed drug, is highly effective for oral ulcers in active BD patients—rapidly reducing ulcer counts, relieving pain, and accelerating healing with good overall safety. It provides a new treatment option for BD patients and represents a major breakthrough in China’s original research drugs for the disease. A phase III clinical trial of mufemilast for BD is ongoing to further verify its long-term efficacy and safety.
Paper link:https://pubmed.ncbi.nlm.nih.gov/41748391/

